Diners at the swanky Atelier Crenn restaurant in San Francisco expect to be served something unusual. After all, the venue boasts three Michelin stars and is widely considered to be one of the world’s top restaurants.
But if all goes according to plan, there will soon be a new dish on the menu that truly is remarkable: chicken that was never part of a living bird.
That peculiar piece of meat – likely to be the first of its kind ever sold in the US – comes from a radical sort of food technology now in development, in which meat is produced by culturing muscle cells in vast tanks of nutrients. A similar effort – to culture mammary cells – is also underway and may soon yield real milk without cows.
The company behind Crenn’s chicken, California-based Upside Foods, got a thumbs-up in November 2022 from the US Food and Drug Administration, which said it had no concerns about the safety of the technology. (The company’s manufacturing facility still requires a certificate of inspection from the US Department of Agriculture.)
This cellular agriculture, as some of its proponents call it, faces formidable technical obstacles before it can ever be more than a curiosity. But if it does reach the mainstream, it offers the prospect of a cruelty-free source of meat and dairy – potentially with a smaller environmental footprint than conventional animal products.
Conceptually, cellular agriculture is straightforward. Technicians take a small tissue sample from a chicken, cow or other animal. From that, they isolate individual cells that go into a bioreactor – basically a big vat of nutrient solution – where the cells multiply manyfold and, eventually, mature into muscle, fat or connective tissue that can be harvested for people to eat.
Products in which these cells are jumbled together, as in ground meat, are easiest to make, and that’s what most cellular meat companies are developing, at least initially. But Upside has a more ambitious goal: to create chicken with whole muscle fibres. “We’ve figured out ways to produce that textural experience,” says Eric Schulze, Upside’s VP of product and regulation. He declines to explain exactly how they do it.
The process takes two to three weeks from start to finish, regardless of whether they are making chicken or beef. That’s much faster than the eight to 10 weeks required to raise a fryer chicken, or the 18 to 36 months needed for a cow. “We’re doing a cow’s worth of meat in 21 days or less,” says Schulze.
One cellular meat product is already available commercially, though not in the US. In Singapore, a few restaurants and street vendors now offer a chicken nugget that contains a mix of cellular meat and plant-based ingredients. The product sells for about the same price as organic, farm-raised chicken, but the true cost of production is higher. “We’re selling it at a loss, for sure,” says Vítor Espírito Santo, senior director of cellular agriculture at Good Meat, the US-based company producing the nugget.
But the cost should come down once the company expands to larger scale, Santo says. “Everything we do right now is more expensive because we are using a 1,200-litre bioreactor. Once we are producing in 250,000 litres, it will be competitive with conventional meat.” The company is now working on gaining approval in the US.
Meat isn’t the only animal product that can come from cell cultures. Several companies are working to produce milk by culturing mammary cells and collecting the milk they secrete. For example, Opalia, a Montreal-based company, grows mammary cells on the surface of a three-dimensional, branched structure that resembles the lobules of a real udder, says CEO Jennifer Côté. The cells secrete milk into the structure’s lobules, where it can be collected and drawn off. Some other companies, such as North Carolina-based BioMilq, are using a similar technology with human mammary cells to produce human breast milk. None are yet on the market.
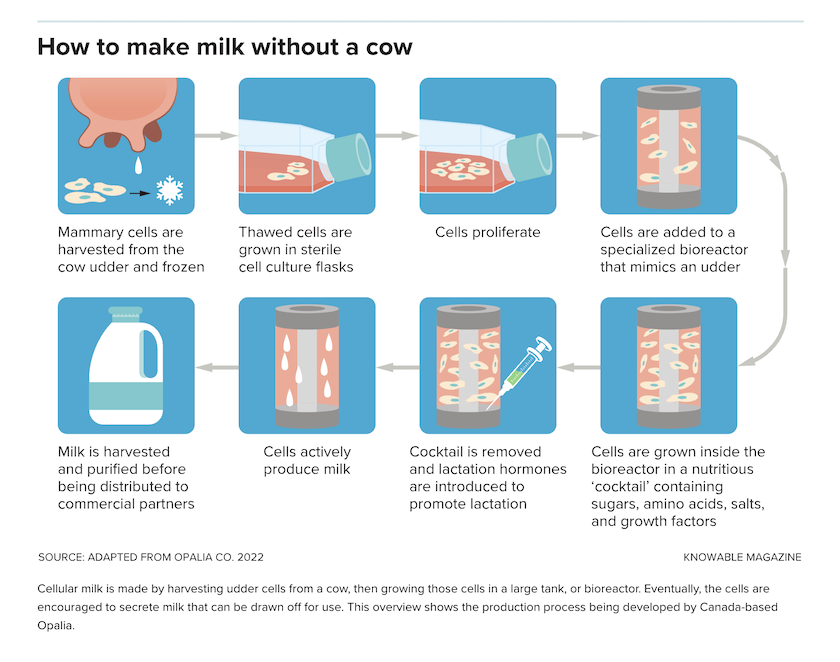
In some ways, the process of making milk is easier than producing meat because the cells themselves don’t need to be harvested and replaced. “The cells we use can stay alive for multiple months on end,” says Côté. That means the company can concentrate on developing cells that secrete a lot of milk, rather than ones that divide rapidly. Moreover, she adds, because the cells themselves are not part of the product, Opalia can genetically modify its cells without the milk itself being a GMO product.
Proponents hope that cellular meat and milk can eventually offer several big advantages over conventional versions. By cutting animals out of the process, cultured products do away with most of the animal-welfare issues that beset modern factory farms. Meat and milk that come from clean culture facilities instead of manure-laden farmyards should also be less likely to carry food-borne diseases, says Elliot Swartz, lead scientist for cultivated meat technology at the Good Food Institute, a Washington DC-based nonprofit organisation supporting alternatives to meat.
Enthusiasts also claim that cell-based products should be more sustainable than conventional animal products because farmers will no longer need to feed, water and house entire animals just to harvest their muscles. It’s hard to know whether this benefit will pan out in reality, since the technology is still under development. Only a few studies have tried to estimate the environmental impact of cell-based meat, and all have made huge assumptions about what future technologies will look like.
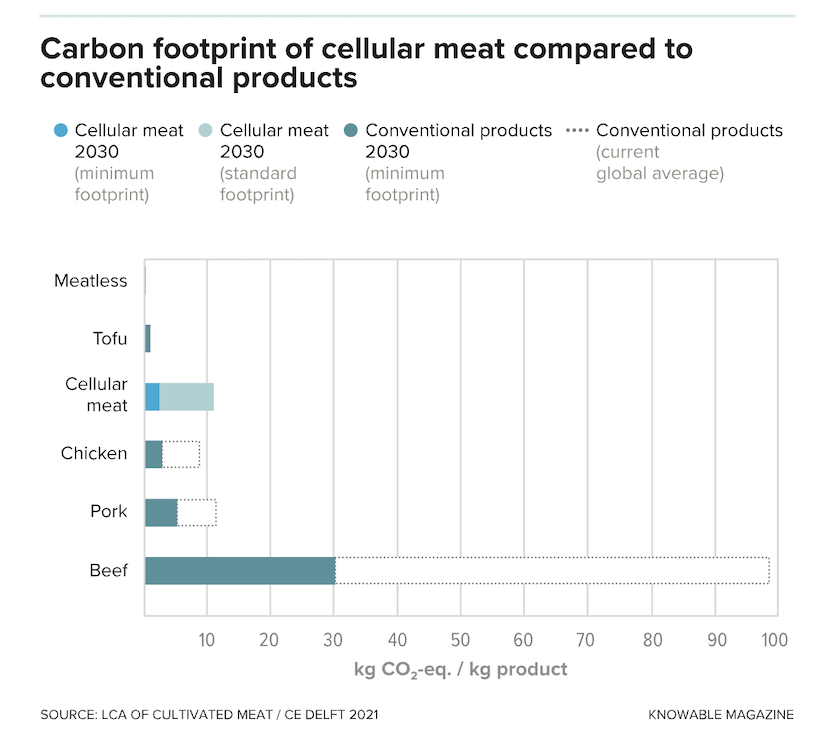
One thing seems clear, however. Cell-based meat relies heavily on electricity for tasks like heating or cooling culture tanks and pumping cells from place to place. If that electricity comes from renewables, the overall carbon footprint of cell-based meat will be much less than if it comes from fossil fuels, says Swartz.
Assuming a relatively green electric grid, though, one careful study of cell-based meat’s potential, by the Dutch consulting company CE Delft, suggests that its environmental footprint is likely to be roughly the same as that of conventional pork or poultry – among the greener conventional meats, by most reckonings – and far less than that of beef.
So far, however, companies and academic researchers have only taken baby steps toward cellular agriculture. If the industry is ever to grow big enough to change the face of global agriculture, it would need to overcome several major hurdles, says David Block, a chemical engineer at the University of California, Davis, who works on the technology behind cultured meat.
One of the biggest challenges, most experts agree, is finding an inexpensive way to supply the nutrients and growth factors the growing cells need. Existing culture media are far too costly and often depend on calves’ blood for molecules such as fibroblast growth factor and insulin-like growth factor 1, which are essential for cell growth and maintenance. Researchers are hoping that relatively unprocessed sources like plant or yeast extract can eventually provide most of the nutrients and vitamins they need, and that they can find a cheaper way to produce the growth factors.
As a step in that direction, Dutch researchers have developed a growth medium using no serum – just off-the-shelf chemicals – to which they add more than a dozen growth factors and other nutrients. Their new medium allowed cow muscle cells to grow almost as well as on calf serum, they reported recently.
Scaling up from research-sized cultures to big commercial operations – an essential step to keeping costs down – may also present problems. The larger the bioreactor, the more difficult it is to ensure that waste products like ammonia are removed, says Ricardo San Martin, a chemical engineer who directs the Alternative Meats Lab at the University of California, Berkeley. Even merely stirring extremely large bioreactors can subject the cells to damaging shear forces, he notes.
The nutrient-supply problem gets even tougher for whole-muscle meats such as steaks or whole chicken breasts. In the animal, such thick slabs of muscle have networks of blood vessels snaking through them, so that every muscle cell is close to a blood supply. Many researchers in the field think replicating that 3D structure in culture poses serious challenges that have yet to be overcome. “I don’t think we are close to growing a steak, and I don’t see it in the next 10 or 15 years,” says San Martin.
Still, proponents remain optimistic that those problems will be settled soon. “Technologically, we’re not concerned,” says Schulze. “With enough time and scientific ingenuity, somebody, somewhere, will find a way to make this work. The cost is the main issue for everyone.”
But cost remains a big stumbling block. The first lab-grown burger patty, produced by a Dutch team in 2013, cost an estimated 250,000 euros (about US$330,000). And while costs have fallen since then, they remain much higher than for conventional meat. In a study that has not yet been peer-reviewed, Block and his colleagues estimated that producing a ground-beef product in a 42,000-litre bioreactor – almost twice as big as the largest in use today for mammalian cells – would cost about $13.80 per pound. To bring the cost down under $6 per pound, only a little pricier than conventional ground beef, would require a much larger, 260,000-litre bioreactor.
But cultured meat may not have to match the price of ground beef or chicken to be commercially viable. Some consumers will probably pay higher prices to avoid the ethical and environmental costs of conventional meat, just as they do today for plant-based meat substitutes like Impossible and Beyond Meat. And some conventional products such as caviar, foie gras or bluefin tuna are so expensive that cultured versions could probably be cost-competitive pretty soon, says Swartz. That would give manufacturers a way to bring in some profits even as they work to bring costs down further.
Another intermediate step could be to use cultured meats to enhance the flavour of plant-based products, as Good Meat is doing now with the part-cultured-meat, part-plant-based meat patties it sells in Singapore. Manufacturers could also add cultured animal fat cells to give a meatier flavour to a plant-based product. “You only need maybe 5 per cent animal fat to achieve that,” says Swartz. Such hybrid products, he thinks, are likely to be the dominant role for cellular meat in the next decade.
Similar first steps could help cultured-milk companies generate revenue before they can match cow’s milk in price. Breast milk offers enough advantages over infant formula, says Swartz, that many consumers are likely to pay high prices for cultured human milk from BioMilq and other companies.
“There are a variety of proteins and fatty acids and sugars that are simply not there if you don’t have breast milk,” says Nurit Argov-Argaman, a lactation physiologist at the Hebrew University of Jerusalem. Argov-Argaman is also chief scientist at Wilk, an Israeli company that is culturing human breast cells to extract high-value components such as fatty acids and lactoferrin, a protein essential to iron uptake, to enrich infant formula.
A few of these cell-cultured meat and milk products should make it to supermarket shelves within the next few years, experts say. But as promising as these first steps are, no one really knows whether cellular meat and milk will eventually grab a significant share of the global market for animal-based foods.
“There are certainly immense challenges – no one’s denying that,” says Schulze. “But our plan is to work on that as an industry. It’s effectively a space race for food. The difference here is we will attempt to rationally solve these challenges one by one in a reasonable time frame – and do it safely, of course, since it’s food.”
About the author: Bob Holmes is a science writer in Edmonton, Alberta, who likes meat but is open-minded about whether or not it comes from an animal.
This article was originally published by Knowable Magazine. Creative Commons Attribution-NoDerivatives 4.0 license (CC BY-ND) 10.1146/knowable-122122-2



